Systems Homeostasis Perspective
This article approaches chronic dysfunction through a systems homeostasis lens, emphasizing stress physiology, neuroendocrine regulation, and downstream tissue tolerance rather than isolated organ-based causality.
A Systems Perspective on Stress, Regulation, and Downstream Dysfunction
It’s increasingly common to hear the phrase: “All health and disease start in the gut.”
This view exists for good reason. The gastrointestinal system is central to digestion, immunity, inflammation, and nutrient assimilation. It is richly innervated, metabolically demanding, and highly responsive to environmental inputs. When systems are under strain, the gut is often one of the first places dysfunction becomes visible.
But visibility is not the same as origin.
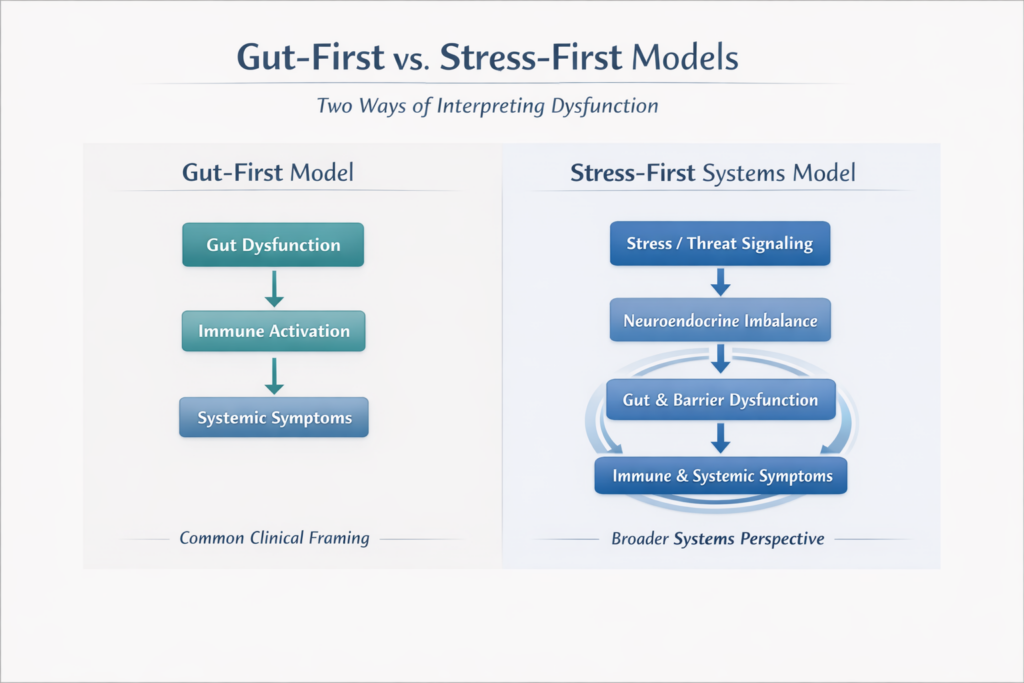
From a systems homeostasis and neuroendocrine perspective, chronic dysfunction rarely begins in the gut. More often, it begins with stress signaling and loss of regulatory control, with gastrointestinal dysfunction emerging downstream.
Why the Gut Often Appears to Be the Starting Point
The gut is uniquely sensitive to systemic stress because it is:
- highly dependent on autonomic balance
- energetically expensive to maintain
- tightly coupled to immune signaling
- responsive to circadian and neuroendocrine regulation
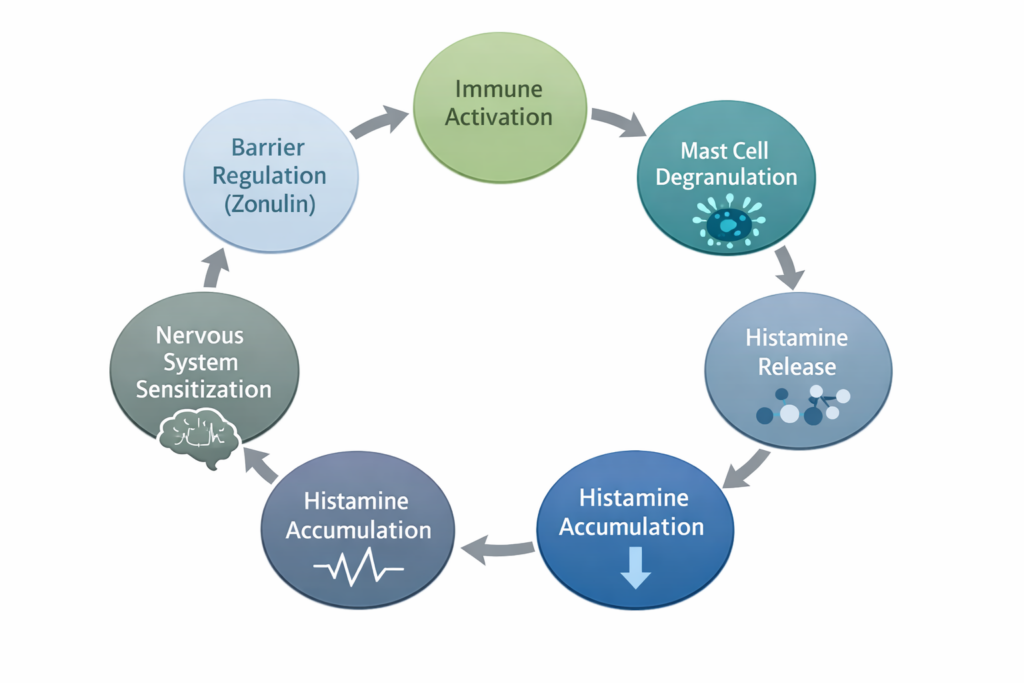
When stress physiology becomes chronic, digestive capacity, motility, barrier integrity, and immune tolerance are among the first functions to degrade.
This makes the gut an excellent early indicator of dysregulation — but not necessarily the primary driver.
Stress as the Upstream Signal
In a systems-based model, stress precedes dysfunction.
Stress here is not limited to psychological stress. It includes:
- perceived threat
- sleep disruption
- metabolic strain
- inflammatory load
- under-recovery
- circadian mismatch
These stressors converge on the HPA axis, autonomic nervous system, and neuroimmune signaling, altering how energy is allocated, how barriers are maintained, and how inflammation is resolved.
As regulatory capacity declines, gastrointestinal function adapts accordingly — often by reducing digestive output, increasing permeability, and activating immune defenses.
When Treating the Gut Alone Falls Short
When dysfunction is assumed to start in the gut, practitioners may:
- over-target gastrointestinal findings
- escalate protocols based on lab abnormalities
- miss upstream drivers of reduced tolerance
- unintentionally increase total system load
In these cases, improving gut markers does not reliably translate into improved clinical outcomes — not because gut work is misguided, but because the system driving the dysfunction has not been addressed.
A More Accurate Systems Sequence
From a neuroendocrine systems perspective, dysfunction more often follows this pattern:
Stress / Threat Signaling
→ Neuroendocrine Dysregulation
→ Autonomic Imbalance
→ Loss of Digestive Capacity
→ Barrier Dysfunction
→ Immune Activation
→ Systemic Symptoms
The gut is central — but it is not primary.
Why This Distinction Matters Clinically
Recognizing the gut as a downstream responder rather than the origin allows care to become:
- better sequenced
- less aggressive
- more tolerable
- more sustainable
Interventions shift from correcting findings to restoring regulatory capacity, often allowing gastrointestinal function to normalize as part of broader recovery.
Systems Reminder
You don’t treat where dysfunction appears.
You treat the system that made dysfunction necessary.
The gut tells an important story — but it is rarely the first chapter.
How I Work
I approach clinical and formulation work through a systems homeostasis framework, prioritizing stress physiology, regulatory capacity, and intervention tolerance before targeting downstream findings. This sequencing supports recovery rather than overwhelming already stressed systems.
Selected References
- Mayer EA et al. The gut–brain axis: interactions between stress, neuroendocrine signaling, and gastrointestinal function. Gastroenterology. https://pubmed.ncbi.nlm.nih.gov/37756251/
- Vanuytsel T et al. Psychological stress and intestinal permeability: a mechanistic review. Gut. https://pmc.ncbi.nlm.nih.gov/articles/PMC10569989/
- Moloney RD et al. Stress, the microbiota–gut–brain axis, and neuroimmune signaling. Neurobiology of Stress. https://www.sciencedirect.com/science/article/pii/S235228952030045X
- Cryan JF, Dinan TG. Mind-altering microorganisms: the gut–brain axis. Nature Reviews Neuroscience. https://www.nature.com/articles/nrn3346