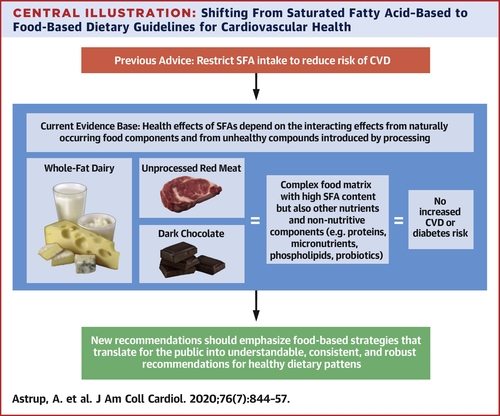
Ingredient Intelligence™ Vol. 1
Vitamin C is one of the most widely used nutrients in modern nutrition, yet the conversation around it has remained surprisingly shallow.
Most discussions stop at dosage or reduce vitamin C to a single molecule — ascorbic acid — without addressing source, biological context, delivery route, or formulation design. In clinical practice and product development, those distinctions often matter far more than the numbers printed on a label.
Vitamin C can function as a daily nutritional requirement, a systems-level regulatory compound, or a therapeutic intervention — depending entirely on how it is sourced, delivered, and paired. Understanding those differences is essential for clinicians, formulators, and companies aiming to work with physiology rather than against it.
Humans Can’t Make Vitamin C — Most Animals Can
Unlike most mammals, humans cannot synthesize vitamin C.
This trait is shared by only a small number of species, most notably humans, other higher primates, and guinea pigs. Most mammals produce vitamin C endogenously in amounts that, when scaled to human body weight, would equate to several grams per day.
As a result, humans are entirely dependent on dietary or supplemental vitamin C and are more sensitive to deficiency during periods of stress, infection, inflammation, or tissue repair. From a systems perspective, vitamin C is foundational, not optional.
Synthetic Vitamin C: What It Is
Most synthetic vitamin C used globally is isolated ascorbic acid produced through an industrial chemical process, often beginning with corn-derived glucose.
Synthetic ascorbic acid is:
- Chemically identical at the molecular level
- Stable, inexpensive, and easy to standardize
This makes it useful for food fortification, short-term correction, and clinical or pharmaceutical applications. However, it represents only one component of how vitamin C appears and functions in nature.
Natural Vitamin C: A Biological Matrix
Whole-food vitamin C sources deliver ascorbic acid within a biological matrix that includes bioflavonoids, polyphenols, and other phytonutrients that influence absorption, tolerance, and cellular signaling.
Common practitioner-grade sources include:
- Acerola cherry
- Camu camu
- Amla (Indian gooseberry)
- Kakadu plum (Australia’s richest known natural vitamin C source)
These cofactors matter because vitamin C does not act in isolation in living systems.
Why Formulation Context Matters
Vitamin C participates in:
- Redox signaling (not just antioxidant activity)
- Collagen synthesis and connective tissue integrity
- Immune cell function
- Endothelial and vascular health
- Iron metabolism
When delivered with its natural cofactors, vitamin C tends to be better tolerated at functional doses and integrates more smoothly into immune and vascular pathways. This helps explain why some individuals experience gastrointestinal irritation with high-dose isolated ascorbic acid, but not with food-based vitamin C systems.
From a formulation standpoint, serious products avoid vitamin C in isolation and instead design systems-level complexes.
Therapeutic Oral Vitamin C and Gastrointestinal Context
Vitamin C has also been used clinically in short-term therapeutic oral protocols, sometimes titrated up to an individual’s bowel tolerance, particularly in gastrointestinal support contexts.
At higher oral doses, unabsorbed vitamin C remains within the intestinal lumen, where it may:
- Increase osmotic activity
- Alter local pH and redox conditions
- Create an environment less favorable for certain pathogens
This approach has historically been used as a temporary therapeutic strategy, distinct from daily nutritional intake. Bowel tolerance varies widely between individuals and reflects differences in gut integrity, inflammatory status, absorptive capacity, and microbial composition.
High-Dose and IV Vitamin C: Context Matters
It is essential to distinguish nutritional vitamin C use from pharmacologic application.
High-dose intravenous (IV) vitamin C has been explored in medical settings such as integrative oncology, where plasma concentrations far exceed what is achievable orally. At these levels, vitamin C may act as a pro-oxidant, generating localized oxidative effects that are context- and dose-dependent.
These applications represent medical therapy, not nutrition, and should not be evaluated by the same criteria as oral vitamin C.
The Real Distinction
This is not a “natural versus synthetic” argument.
It is a context and intent argument.
- Synthetic ascorbic acid has a role in fortification and medical therapy
- Natural vitamin C systems are better aligned with long-term immune, vascular, and metabolic support
The real question is not how much vitamin C — but in what form, with what cofactors, and for what purpose.
That’s the vitamin C conversation we’re not having — and the one that matters most.
Vitamin C is one of the most widely used nutrients in modern nutrition, yet the conversation around it has remained surprisingly shallow.
Most discussions stop at dosage or reduce vitamin C to a single molecule — ascorbic acid — without addressing source, biological context, delivery route, or formulation design. In clinical practice and product development, those distinctions often matter far more than the numbers printed on a label.
Vitamin C can function as a daily nutritional requirement, a systems-level regulatory compound, or a therapeutic intervention — depending entirely on how it is sourced, delivered, and paired. Understanding those differences is essential for clinicians, formulators, and companies aiming to work with physiology rather than against it.
Humans Can’t Make Vitamin C — Most Animals Can
Unlike most mammals, humans cannot synthesize vitamin C.
This trait is shared by only a small number of species, most notably humans, other higher primates, and guinea pigs. Most mammals produce vitamin C endogenously in amounts that, when scaled to human body weight, would equate to several grams per day.
As a result, humans are entirely dependent on dietary or supplemental vitamin C and are more sensitive to deficiency during periods of stress, infection, inflammation, or tissue repair. From a systems perspective, vitamin C is foundational, not optional.
Synthetic Vitamin C: What It Is
Most synthetic vitamin C used globally is isolated ascorbic acid produced through an industrial chemical process, often beginning with corn-derived glucose.
Synthetic ascorbic acid is:
- Chemically identical at the molecular level
- Stable, inexpensive, and easy to standardize
This makes it useful for food fortification, short-term correction, and clinical or pharmaceutical applications. However, it represents only one component of how vitamin C appears and functions in nature.
Natural Vitamin C: A Biological Matrix
Whole-food vitamin C sources deliver ascorbic acid within a biological matrix that includes bioflavonoids, polyphenols, and other phytonutrients that influence absorption, tolerance, and cellular signaling.
Common practitioner-grade sources include:
- Acerola cherry
- Camu camu
- Amla (Indian gooseberry)
- Kakadu plum (Australia’s richest known natural vitamin C source)
These cofactors matter because vitamin C does not act in isolation in living systems.
Why Formulation Context Matters
Vitamin C participates in:
- Redox signaling (not just antioxidant activity)
- Collagen synthesis and connective tissue integrity
- Immune cell function
- Endothelial and vascular health
- Iron metabolism
When delivered with its natural cofactors, vitamin C tends to be better tolerated at functional doses and integrates more smoothly into immune and vascular pathways. This helps explain why some individuals experience gastrointestinal irritation with high-dose isolated ascorbic acid, but not with food-based vitamin C systems.
From a formulation standpoint, serious products avoid vitamin C in isolation and instead design systems-level complexes.
Therapeutic Oral Vitamin C and Gastrointestinal Context
Vitamin C has also been used clinically in short-term therapeutic oral protocols, sometimes titrated up to an individual’s bowel tolerance, particularly in gastrointestinal support contexts.
At higher oral doses, unabsorbed vitamin C remains within the intestinal lumen, where it may:
- Increase osmotic activity
- Alter local pH and redox conditions
- Create an environment less favorable for certain pathogens
This approach has historically been used as a temporary therapeutic strategy, distinct from daily nutritional intake. Bowel tolerance varies widely between individuals and reflects differences in gut integrity, inflammatory status, absorptive capacity, and microbial composition.
High-Dose and IV Vitamin C: Context Matters
It is essential to distinguish nutritional vitamin C use from pharmacologic application.
High-dose intravenous (IV) vitamin C has been explored in medical settings such as integrative oncology, where plasma concentrations far exceed what is achievable orally. At these levels, vitamin C may act as a pro-oxidant, generating localized oxidative effects that are context- and dose-dependent.
These applications represent medical therapy, not nutrition, and should not be evaluated by the same criteria as oral vitamin C.
The Real Distinction
This is not a “natural versus synthetic” argument.
It is a context and intent argument.
- Synthetic ascorbic acid has a role in fortification and medical therapy
- Natural vitamin C systems are better aligned with long-term immune, vascular, and metabolic support
The real question is not how much vitamin C — but in what form, with what cofactors, and for what purpose.
That’s the vitamin C conversation we’re not having — and the one that matters most.
Canonical CTA
If you are a clinic or practitioner group looking to integrate systems-based immune, cardiovascular, or metabolic support, I work with clinics to design non-pharmacologic, physiology-informed strategies aligned with real-world practice.
If you are a company developing practitioner-grade supplements, powders, or functional drinks, I provide formulation strategy, ingredient architecture, and product development support from concept through commercialization.
Clinical support: OptimumHealthConsulting.com
Formulation consulting: HealthspanFormulations.com
#ProductFormulation #FunctionalNutrition #VitaminC #NutraceuticalInnovation #IngredientIntelligence #RobLamberton #RobertLamberton